From Validation to Governance: NAMs, EHDS, and the Structural Redefinition of Regulatory Evidence in the EU
EU Health Systems · Regulation · Translation
EDITION 24 · APRIL 2026
THE SYSTEM CHALLENGE
In March 2026, the FDA issued draft guidance on New Approach Methodologies (NAMs). It formalises a clear shift: validation is no longer defined by a single method, but by context of use, biological relevance, and fit-for-purpose performance.
This is not a technical update. It changes how regulation itself works.
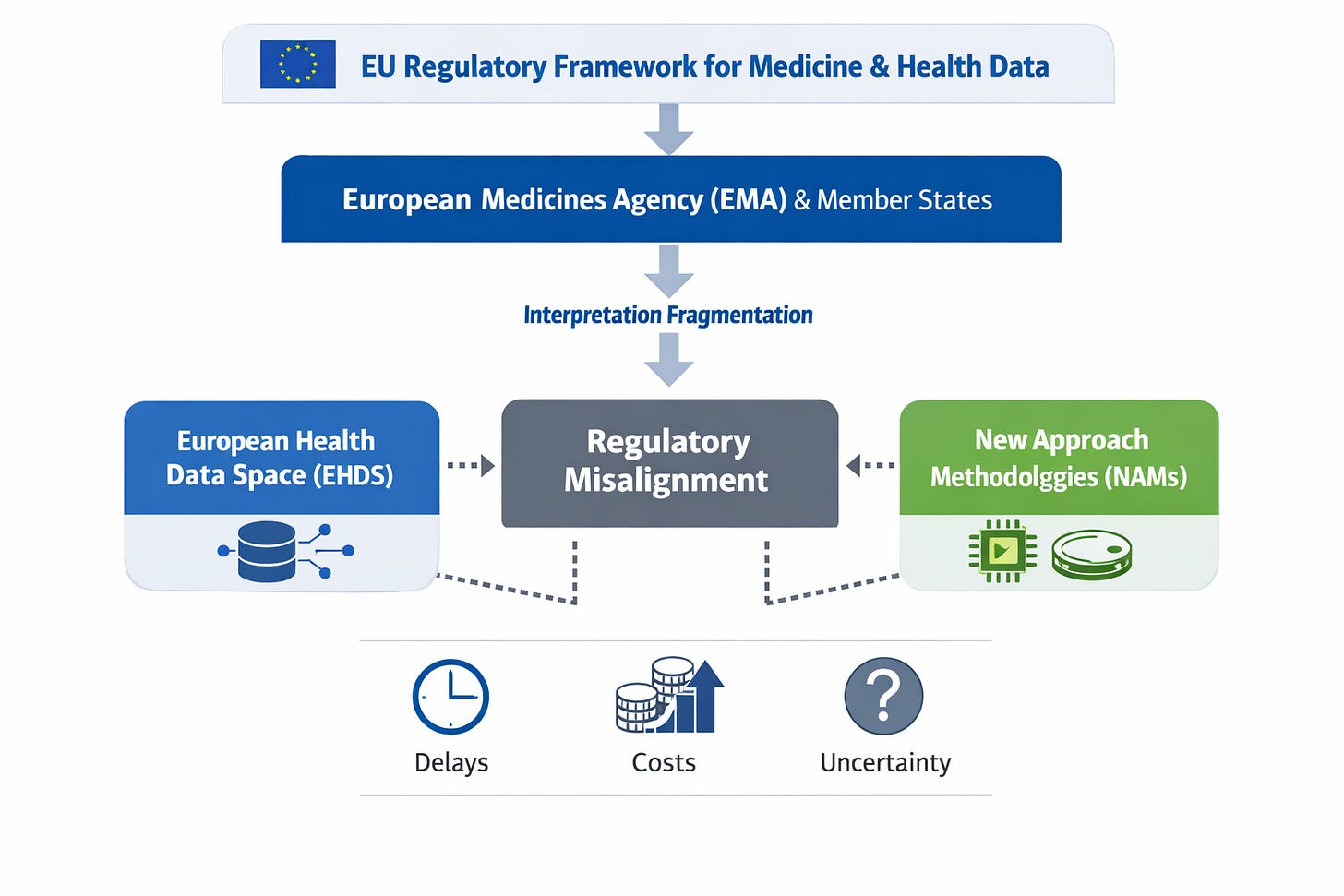
For the European Union, this arrives at a sensitive moment. The European Medicines Agency coordinates regulation across 27 Member States, while the European Health Data Space (EHDS) is still being implemented. At the same time, there is no fully shared framework for how NAM-based evidence should be interpreted.
So the system is now in a mismatch: a regulatory structure built for standardised evidence is being asked to evaluate context-dependent evidence.
SCALE
The impact becomes clearer when looking at the numbers.
EU pharmaceutical development cycles: 8–12 years
Nonclinical R&D: 20–35% of total preclinical costs
Programme cost range (oncology, neurology): €1.5–3.5 billion
Validation-related delays: 6–18 months at key decision points
Cost increase from regulatory variability: 15–25% per programme
These are not marginal inefficiencies. They accumulate across the entire development pipeline, especially in oncology and CNS, where uncertainty is already high.
TWO STRUCTURAL CAUSES
1. Limits of traditional models
Animal models and standard in vitro systems are increasingly unable to predict human outcomes in complex diseases, especially in neurology, neurophysiology and oncology.
This is widely recognised.
As a result, New Approach Methodologies (NAMs) — including computational models, organ-on-chip systems, and hybrid frameworks — are no longer experimental. They are now part of regulatory submissions.
But they introduce a new requirement: interpretation depends on context, not fixed benchmarks.
That is where the gap appears. Existing EMA-aligned systems were not designed for this type of evidence logic.
2. Fragmented interpretation across Member States
The EMA provides coordination, but interpretation still varies across countries.
For the same NAM dataset, different national authorities may reach different conclusions depending on how they interpret performance metrics like sensitivity, specificity, and predictive validity.
This is not a failure of institutions. It is a structural limitation: regulation is harmonised, but interpretation is not.
THE EHDS CONNECTION
The European Health Data Space adds another layer.
It is designed to enable secondary use of health data across Europe — including research, drug development, and regulatory evaluation.
But if NAM interpretation remains inconsistent, EHDS data will not be assessed in a uniform way.
In other words: the infrastructure becomes shared, but the interpretation of its outputs does not.
These two processes — NAMs evolution and EHDS implementation — are moving in parallel, but not yet in coordination.
That gap is becoming operational.
REGULATORY CONSEQUENCE
If interpretation of NAM evidence is not harmonised, three effects follow:
Regulatory workload increases by 10–20% due to duplicated assessments across Member States
Predictability decreases for pharmaceutical companies working under EMA coordination
Investment becomes less attractive in CNS and oncology compared to regions with clearer frameworks
The shift toward context-based validation is not optional — it is already happening. The question is whether the EU can implement it coherently.
TWO STRUCTURAL DIRECTIONS
1. EU-level harmonisation of NAM interpretation
A coordinated framework under EMA guidance, ideally operational by 2027, would align how context-of-use validation is applied across Member States.
Estimated effect:
10–15% reduction in regulatory duplication
stronger predictability across jurisdictions
This requires treating NAM interpretation as a shared EU coordination priority, not a national discretion challenge.
2. Standardised reporting of predictive performance
All NAM-based submissions should use a common format for reporting:
sensitivity
specificity
predictive validity
This is not a new regulatory barrier. It is a way to make results comparable across systems.
It reduces interpretation differences without limiting innovation.
STRUCTURAL ASSESSMENT
The EU is entering a transition where science is moving faster than regulatory alignment.
NAMs and EHDS are both advancing, but without full coordination between them.
This creates a measurable gap between data generation and regulatory interpretation.
That gap already affects timelines, costs, and investment decisions.
The Commission and EMA have the mandate to close it. The key question is timing — whether alignment happens early, or only after fragmentation becomes structural.
NeuroEdge Nexus translates neuroscience, AI, and European regulatory frameworks into decision-grade strategic analysis. Season 2 (2026) focuses on governance, infrastructure coordination, and the implementation gap in digital brain health.