The Body as Data: How Digital Biomarkers Are Rewriting the Rules of Brain Health
How Wearable Devices, AI Models, and Digital Biomarkers Are Transforming Clinical Trials, Prevention, and Precision Medicine
NeuroEdge Nexus — Edition 21 | March 2025
A brain does not malfunction on the day of diagnosis. In clinical and research settings, epileptiform activity detected on EEG, disrupted REM sleep architecture, and subtle gait alterations have each been observed in patients years before a formal diagnosis of neurodegeneration. In animal models of Alzheimer’s disease, epileptiform activity has been observed to precede cognitive decline. Emerging human studies suggest that continuous physiological monitoring may help detect similar early network alterations. The signal was always present. Only now are we building the instruments capable of reading it systematically.
Recent advances in digital biomarkers, wearable devices, artificial intelligence, and decentralized clinical trials are beginning to change how biological signals can be observed and interpreted. Instead of relying solely on periodic clinical measurements, researchers are increasingly exploring continuous physiological monitoring as a potential foundation for future precision medicine.
At the center of this transition is a growing research field focused on digital biomarkers — and the clinical, regulatory, and ethical infrastructure required to translate them into practice.
Digital Biomarkers: Measuring Physiology Beyond the Clinic
Digital biomarkers are objective, quantifiable physiological or behavioral data collected through digital technologies such as wearable sensors, mobile devices, and home monitoring systems. Unlike traditional biomarkers derived from blood samples or imaging tests, digital biomarkers capture physiological signals generated during everyday life.
Examples under active investigation in clinical research include:
• Sleep architecture patterns and REM sleep disruption
• Gait and motor activity changes
• Speech and cognitive behavior signals
• Heart rate variability and cardiovascular dynamics
• Epileptiform and neurophysiological signals captured through portable EEG
Because these signals can be collected continuously, they provide a longitudinal perspective on human physiology that cannot be captured through occasional clinical measurements alone. This capability is particularly relevant for neurological disorders, where disease processes often develop gradually over many years — and where the gap between biological signal and clinical diagnosis may be measured in years, not months.
Examples of Digital Biomarkers Under Investigation
Several types of digital biomarkers are currently being studied in neurodegenerative disease research.
Eye-tracking metrics
Subtle changes in eye movement patterns — including saccades, pupil dynamics, and blink rate — may reflect early alterations in neural circuits involved in attention and motor control.
Gait and motor activity patterns
Wearable sensors can detect small changes in gait symmetry, walking speed, and motor coordination that may precede clinical diagnosis in disorders such as Parkinson’s disease.
Sleep-based biomarkers
Sleep monitoring technologies are increasingly used to analyze sleep architecture and neural oscillations. Changes in non-REM sleep spindles and sleep fragmentation have been associated with neurodegenerative processes in several studies. Research has also highlighted the role of sleep in metabolite clearance from the brain — a mechanism with direct relevance to neurodegeneration.
Wearable Devices and Continuous Health Monitoring
Wearable health technologies have expanded the ability to monitor physiological signals outside traditional healthcare environments. Many wearable systems can measure activity levels, sleep cycles, cardiovascular dynamics, and physiological rhythms during daily life. Newer research platforms are also exploring wearable technologies capable of capturing neurophysiological signals, including portable EEG and other biosignal monitoring tools.
These systems generate large datasets describing physiological processes over time. The scientific value of these datasets lies not in any single measurement, but in the patterns that emerge across time
— patterns that conventional episodic testing is structurally unable to detect.
Artificial Intelligence and Digital Biomarker Discovery
Continuous physiological monitoring generates large volumes of multidimensional data. Artificial intelligence and machine learning techniques are increasingly applied in research settings to analyze these datasets. A comprehensive scoping review published in NPJ Digital Medicine in 2025 identified 86 AI models applied to digital biomarkers in Alzheimer’s disease research alone — with models distinguishing Alzheimer’s patients from healthy controls achieving an average AUC of 0.887, a level of predictive performance that illustrates the analytical potential of these datasets, while also highlighting the need for rigorous external validation.
AI models can integrate multiple sources of information — wearable sensor data, behavioral signals, physiological measurements, and clinical variables — to identify digital biomarker signatures that may correlate with disease risk, progression, or therapeutic response. Such approaches are currently being explored across neurodegenerative diseases, sleep disorders, movement disorders, and mental health conditions.
Digital Biomarkers and Decentralized Clinical Trials
The development of wearable monitoring technologies has contributed to the emergence of decentralized clinical trials. In these studies, participants can be monitored remotely using digital devices rather than attending frequent hospital visits. Continuous physiological data collected in real-world environments may provide additional insights into disease progression and treatment response.
Digital biomarkers derived from these datasets can serve as digital endpoints
— offering new ways to measure outcomes in clinical research that are more sensitive, more continuous, and more ecologically valid than periodic clinical assessments.
This is particularly relevant in neurological research, where symptoms fluctuate and evolve gradually.
Precision Medicine and Individual Health Trajectories
The broader objective of digital biomarker research is to support the development of precision medicine — approaches that tailor prevention strategies, diagnostics, and treatments to the individual biological characteristics of each patient. Continuous monitoring of physiological signals may contribute to this goal by enabling earlier identification of disease-related changes, more detailed tracking of disease progression, individualized monitoring of treatment responses, and improved understanding of patient-specific health trajectories.
In neurological disorders, where subtle physiological changes may occur long before symptoms appear — as both animal model research and longitudinal clinical observation suggest — such approaches represent a fundamental shift in how we define the onset of disease.
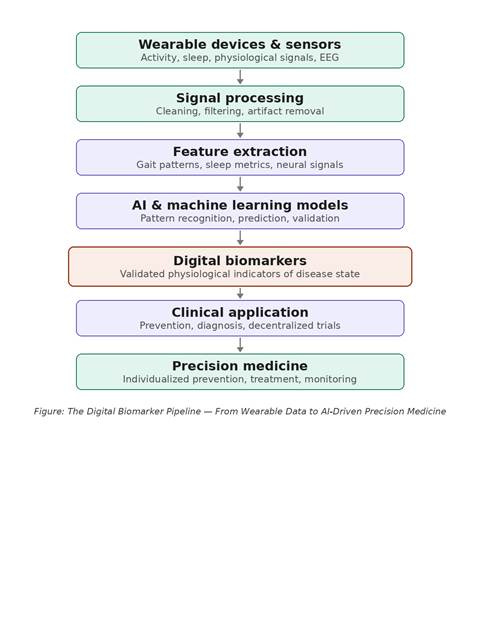
The Digital Biomarker Pipeline
Digital biomarkers do not emerge directly from raw sensor data. They result from a structured analytical process that transforms physiological signals into interpretable clinical indicators.
Figure: The Digital Biomarker Pipeline — From Wearable Data to AI-Driven Precision Medicine
Wearable devices first capture physiological signals such as activity patterns, sleep dynamics, neurophysiological recordings, and biosignals. These data are processed through signal-cleaning and preprocessing pipelines to remove noise and artifacts. From these processed signals, researchers extract physiological features — gait patterns, sleep metrics, EEG characteristics, or speech markers. Artificial intelligence models then analyze these features to identify patterns associated with disease states or health outcomes. When validated in research and clinical settings, these patterns may serve as digital biomarkers, supporting new approaches to prevention, diagnosis, and monitoring.
The Real Bottleneck: Not Technology, But Infrastructure
The scientific and technological foundations of digital biomarker research are maturing rapidly. The instruments exist. The AI models are being developed. The clinical signals — in sleep, in gait, in neurophysiology — are increasingly well-characterized.
The primary bottleneck today is not capability. It is the absence of robust clinical, regulatory, and organisational infrastructure to translate these signals into validated, deployable tools. What the field currently lacks is standardised agreement on what constitutes a validated digital endpoint — the kind of regulatory clarity that would allow digital biomarkers to move from research observation to clinical and pharmaceutical application with confidence.
The challenge now facing digital biomarker research is not primarily technological. The instruments capable of capturing physiological signals at scale already exist. What remains unresolved is the scientific and regulatory framework required to transform those signals into clinically validated endpoints. Without standardisation in signal processing, validation protocols, and regulatory acceptance, digital biomarkers risk remaining powerful research tools that never fully translate into routine clinical practice.
Governance, Regulation, and Human Rights
The integration of continuous physiological monitoring into healthcare raises important governance and regulatory questions. Digital biomarkers involve the collection and analysis of highly sensitive personal data, including behavioral and neurophysiological signals collected during daily life.
In the European Union, regulatory frameworks are actively evolving to address these challenges. The European Health Data Space, the Artificial Intelligence Act, and the General Data Protection Regulation collectively define how health data and AI technologies can be used responsibly. These frameworks are not obstacles to innovation — they are the conditions under which responsible innovation becomes scalable and trustworthy.
A Changing Paradigm in Medicine
Medicine has traditionally relied on occasional clinical measurements to observe biological systems. Digital biomarkers, wearable monitoring technologies, and AI-driven analytics are beginning to enable a different approach — one based on continuous observation of physiological signals across time and daily life.
For neuroscience and brain health research, where complex network dynamics evolve slowly and subtly — where epileptiform signals, sleep disruption, and gait changes may precede diagnosis by years — this transition is not incremental. It is a fundamental reframing of when disease begins, and therefore when intervention becomes possible.
Progress will depend not only on scientific and technological advances, but on the creation of the clinical, regulatory, and ethical infrastructure that allows these tools to reach the patients who need them most.
References
Qi W et al. Alzheimer’s disease digital biomarkers multidimensional landscape and AI model scoping review. NPJ Digital Medicine. 2025;8:366. doi:10.1038/s41746-025-01640-z
Levendowski DJ et al. Proof-of-concept for characterization of neurodegenerative disorders utilizing two non-REM sleep biomarkers. Frontiers in Neurology. 2023;14:1272369. doi:10.3389/fneur.2023.1272369
Xie L et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342(6156):373-377. doi:10.1126/science.1241224
NeuroEdge Nexus translates neuroscience, AI, and European regulatory frameworks into strategic analysis. Season 2 (2026) examines governance implementation, neurological rights, and the translation of regulatory mandate into functional infrastructure.
This analysis represents expert commentary on neurological rights and brain data governance. It is not legal advice. Organizations implementing neuroscience systems should consult appropriate legal and ethics specialists.